What it is
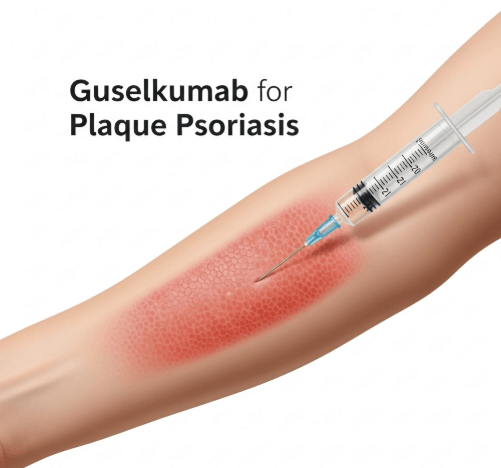
➝ Guselkumab is a biologic therapy used for the treatment of moderate-to-severe plaque psoriasis.
➝ It is a human monoclonal antibody that specifically targets interleukin-23 (IL-23), a key cytokine in the inflammatory pathway of psoriasis.
➝ By blocking IL-23, guselkumab reduces the overactive immune response that causes skin cell overgrowth, scaling, redness, and itching.
➝ Unlike older systemic drugs such as methotrexate or cyclosporine, guselkumab offers highly targeted treatment with fewer risks of organ toxicity.
Why it’s done
→ For patients with moderate-to-severe plaque psoriasis who do not respond well to topical treatments or systemic oral medications.
→ Helps clear stubborn plaques on the scalp, nails, and body that are difficult to treat.
→ Reduces psoriasis-related itching, discomfort, and scaling, improving quality of life.
→ Often prescribed to patients who need long-term, safe, and effective control.
→ In Korea, guselkumab is commonly used in advanced dermatology clinics for patients seeking biologic therapies with strong efficacy.
Alternatives
→ Other biologics:
- IL-17 inhibitors (e.g., secukinumab, ixekizumab).
- IL-23 inhibitors (e.g., risankizumab, tildrakizumab).
- TNF-alpha inhibitors (e.g., adalimumab, etanercept).
→ Oral systemic drugs: Methotrexate, cyclosporine, acitretin, apremilast.
→ Phototherapy: Narrowband UVB or PUVA, often for milder cases or in combination with systemic drugs.
→ Topical treatments: Steroids, vitamin D analogs, coal tar, calcineurin inhibitors (for localized or mild cases).
Preparation
→ Patients undergo baseline tests before starting guselkumab:
- Tuberculosis screening.
- Hepatitis B and C tests.
- Complete blood count and liver function tests.
→ Any active infections must be treated before therapy begins.
→ Vaccination history is reviewed, since live vaccines are not recommended during biologic therapy.
→ Patients are counseled about injection training, as guselkumab is self-administered at home after the initial doses.
How it’s Done
→ Guselkumab is administered as a subcutaneous injection (under the skin).
→ Dosing schedule:
- Initial injection at week 0.
- Second injection at week 4.
- Maintenance injections every 8 weeks thereafter.
→ It is usually injected in the thigh, upper arm, or abdomen.
→ Many patients appreciate guselkumab’s convenient dosing schedule compared to biologics that require more frequent injections.
Recovery
→ Patients often notice improvement within 2–4 weeks of starting therapy.
→ Significant plaque clearance usually occurs by 12–16 weeks.
→ Many patients achieve PASI 90 or even PASI 100 responses (90–100% clearance of skin lesions).
→ Improvement in itching, scaling, redness, and nail psoriasis enhances comfort and confidence.
→ Long-term studies show sustained effectiveness with continued injections every 8 weeks.
Complications
→ Common mild side effects: Injection site reactions, headaches, upper respiratory infections.
→ Less common but possible: Joint pain, diarrhea, fatigue.
→ Serious risks: Increased risk of infections (though lower compared to older biologics).
→ Contraindications: Patients with active infections, tuberculosis, or immune-compromised conditions.
→ Long-term safety data is favorable, with guselkumab considered one of the safer biologics for psoriasis.
Treatment Options in Korea
→ Korean dermatology clinics and university hospitals are highly experienced in using guselkumab and other biologics.
→ It is commonly offered to patients with severe, widespread, or treatment-resistant plaque psoriasis.
→ Korean doctors often integrate guselkumab into personalized care programs, including topical treatments, moisturization therapy, and lifestyle guidance.
→ Monitoring is carried out regularly, though guselkumab requires less frequent lab testing than methotrexate or cyclosporine.
→ In Korea, guselkumab is valued for its convenience (8-week schedule), high success rate, and excellent safety profile, making it a leading choice for long-term plaque psoriasis management.